Integrated compounding quality control
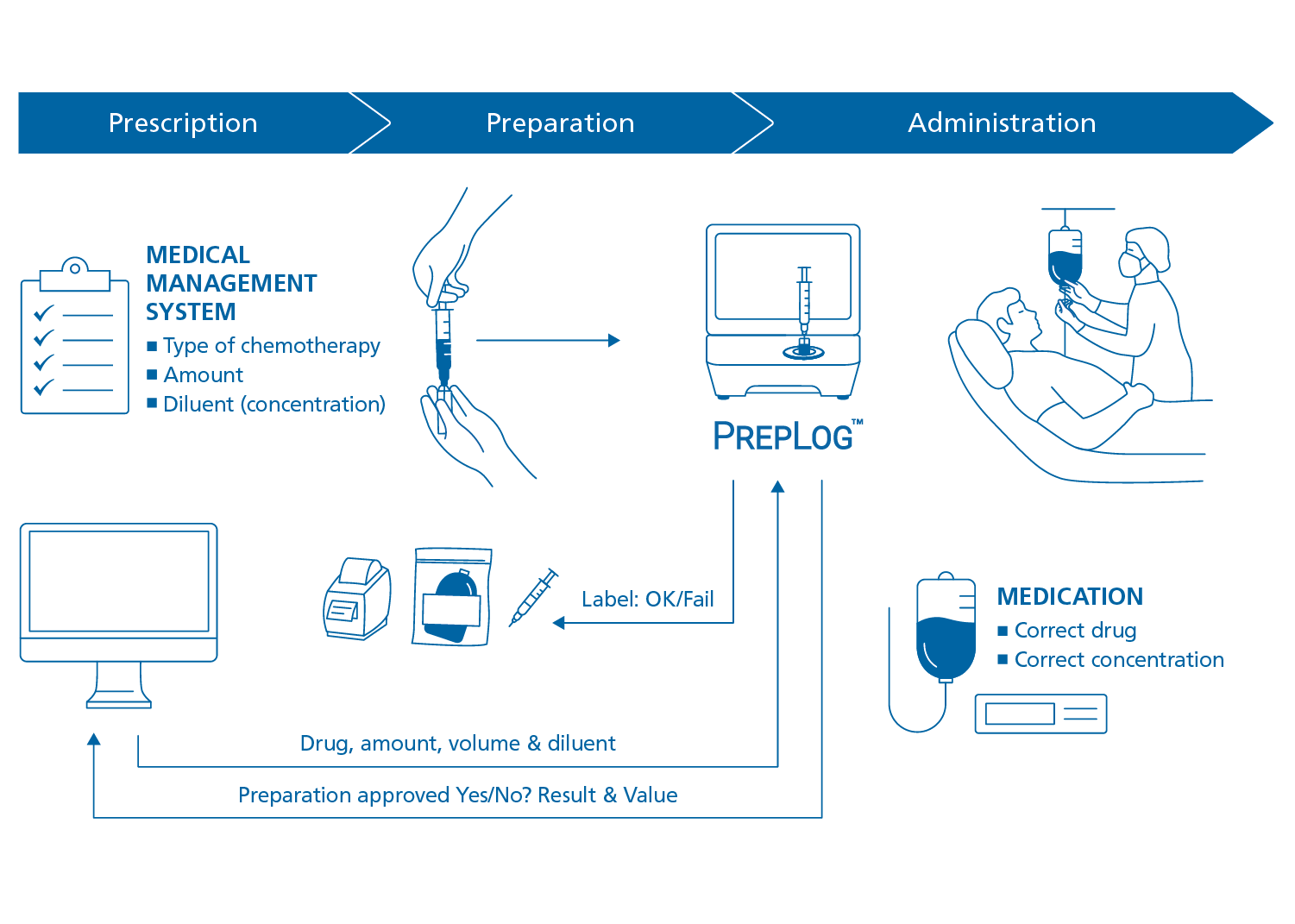
Tools that help hospital staff feel confident about the medication they prepare not only reduce the risk for error, but also lower stress and help avoid wasting correctly prepared medication due to uncertainty. PrepLog™ provides that level of confidence. An integrated solution compatible with a range of different Medication Management Systems (MMS), the PrepLog™ platform receives requests for testing from the MMS so that quality control of drug preparations can be performed and reported in an automated way.
Integrate prescription software with compounding quality control
PrepLog™ is an innovative, unique product workflow solution based on patented DrugLog™ technology. PrepLog™ is a comprehensive system that integrates Prescription Software or MMS to ensure safe and reliable drug preparations for patients. The integrated PrepLog™ solution automatically receives information about the expected substance and concentration from the prescription system or MMS. The result after the completed measurement is also sent back to the electronic prescription system, MMS or patient record. Underpinning all of this is the well-proven DrugLog™ technology, which guarantees fast and reliable measurements.
Thanks to PrepLog™, patients receive the intended medication – and nothing else.
Key features of PrepLog™
Fast, accurate validation
PrepLog™ offers an easy-to-use method for true validation of each chemotherapy dose that is sent to the oncology ward. It connects to local networks which integrate with the Prescription Software of choice, MMS or patient record.
How it works
1. PrepLog™ receives measurement request and builds a work list.
2. Withdraw a small sample (0.3–0.5ml) from the compounded medication, inject into a cuvette and insert into PrepLog™.
3. PrepLog™ verifies that the medication is the correct prescribed drug and correct concentration.
4. Print a label and attach to the medication bag or syringe. The test result is sent to the prescription system for full traceability.
Why PrepLog™?
Documentation

PrepLog™
Brochure
- Europe
Download PDF

Pharmacolog Dashboard™
Brochure
Download PDF

Customer Story -
L’Hôpital Privé du Confluent, Nantes, France
Download PDF

Pharmacolog
Service & Support
Brochure
Download PDF